www.know_the_risk_of_5fu_chemotherapy.com
Insist on DPYD Testing before Treatment: it is the New standard of care!
Last Update: 7 Dec 2025
This year, 2025, has seen major steps in ensuring the safe use of 5-FU and Xeloda chemotherapy though it is clear more work is necessary to ensure universal testing for DPD deficiency.
Authorities responsible for treatment guidelines and drug labels have finally proclaimed that testing patients for DPD deficiency should happen before treating patients with fluoropyrimidines.
The US Food and Drug Administration issued a Black Box warning for Xeloda (capecitabine) that reads:
The National Comprehensive Cancer Network's (NCCN) colon cancer panel updated its treatment guidelines by pointing out the FDA's Box Warning.
Yet despite these recent changes (October 2025), we are finding that patients are receiving treatment and experiencing toxicities without first having the option to test for DPD deficiency to determine if treatment is safe for them.
Before you or a loved one consider chemotherapy with these drugs, you should absolutely order a genetic test (of the DPYD gene). If your oncologist dismisses the idea, you should make them aware of the newest guidelines or consider another physician.
For information on clinics leading the way in testing and on test labs, visit Test4DPD.org
Date of latest revision: 19 March 2025
UNDERSTAND the RISK BEFORE STARTING TREATMENT
If you have been diagnosed with stage III or IV breast, head/neck, colon, rectal, or anal cancer, chances are your doctors are discussing with you the need for chemotherapy. The
standard drug for treating these types of cancers is fluorouracil (popularly known as 5-FU); capecitabine (Xeloda) may also be prescribed.
Oncologists have used these drugs for many years in treating cancers and the perception is that they are "fairly well tolerated" by patients.
Wrong! In fact a significant number of patients (between 10% -- 40% of treated patients) suffer severe toxic reactions that lead to costly hospital stays and over 1,000 patients will DIE ANNUALLY in the US.
5-FU and capecitabine depend on the patient's body to metabolize the drug quickly to prevent it from damaging healthy cells. This metabolic process is critically dependent on an enzyme: dihydropyrimidine dehydrogenase (aka DPD). When a patient's DPD activity is compromised, 5-FU and capecitabine destroy healthy cells and put a person at grave risk. DPD deficiency presents a single point of failure.
Most people are unaware of this condition until too late: do not wait until you suffer an adverse reaction. Insist that your oncologist test for the deficiency before starting treatment.
Tests are available to determine if a person has DPD deficiency (see https://test4dpd.org/why-test/) and some cancer centers/clinicians are pre-screening even in the absence of national guidelines (see leaders).
You should insist on discussing with your oncologist ways to minimize your risk and to provide you a better outcome before you start
chemotherapy.
Purpose
The goal of this site is to encourage patients preparing for chemotherapy with 5-FU/capecitabine to discuss personalized chemotherapy regimens with their oncologists before starting treatment. You should not take at face value that the treatment is "fairly well tolerated".
Fluorouracil (also known as 5-FU) and Capecitabine (or Xeloda) have been used for many years as a key chemotherapy agent in the treatment of patients with colon, rectal, anal cancers and is sometimes used to treat head/neck and breast cancer patients. Oncologists consider this treatment an effective and low risk means of destroying cancerous cells.
However, medical journals have for years pointed to a greater risk of toxic reactions that can lead to hospitalization and even to death (1,000 or more fatalities annually in the US).
The intent here is NOT to scare patients, facing cancer is frightening enough, nor to discourage patients from seeking professional medical care. Instead, the intent is to increase awareness of the risk and to embolden patients and their families to have an educated discussion with their physicians about the risks associated with 5-FU chemotherapy.
So the following information may prepare you to have a frank discussion with your care provider:
- Why Test? (external link)
- Warning Signs to Report & Act Upon
Dedication
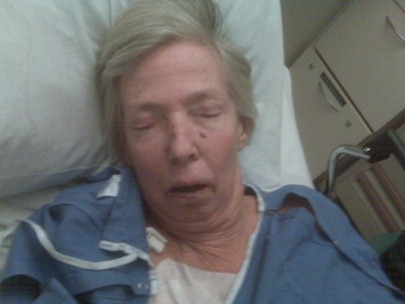
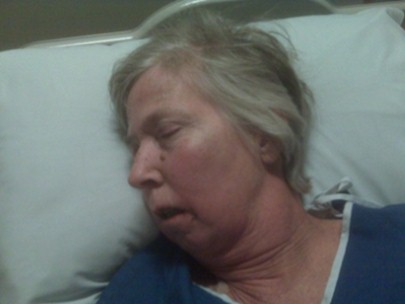
Kathryn received one round of FOLFOX (5-FU, leucovorin, and oxaliplatin) and passed away within 3 weeks (see Personal Stories). Kathryn inspired many people in our community in many ways and here her inspiration takes form among these pages in the hope that it will achieve one of her last wishes:
"I am optimistic and very grateful to all who have traveled this path before me and contributed to the knowledge and experience of those whose vocation is to heal people like me. May my experience also contribute to the body of learning."
This site is dedicated to honor her wish and to help others avoid the fate she suffered.